MYMT2019
Takahiro Matsui, Takuma Yagasaki, Masakazu Matsumoto, and Hideki Tanaka
Phase diagram of ice polymorphs under negative pressure considering the limits of mechanical stability
J. Chem. Phys. 150, 041102 (2019)
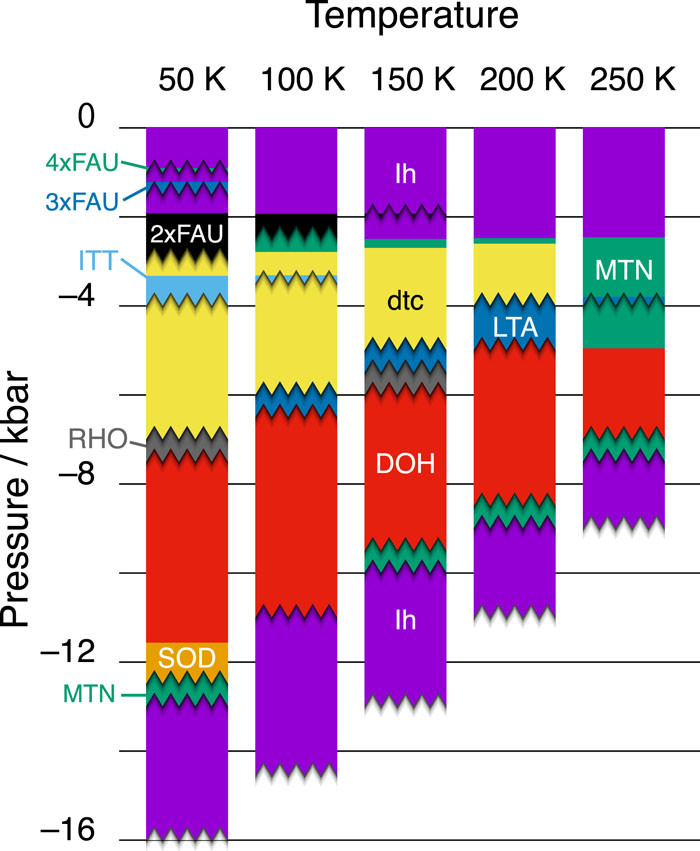
負の圧力領域(物質を伸張した状態)の最安定相は気相ですが、結晶も準安定状態として存在することができます。水については、2014年に通常の氷よりも密度の低い結晶氷XVIが特殊な製法により作られ、負圧で安定となることが予想されたため、新たな低密度氷を探しだすことに注目が集まっています。我々は、以前の論文(MHYMT2017)で、負圧で安定となる結晶構造は無数に存在し、なかでもデザインされた氷aeroiceは極めて広い温度圧力範囲で安定であることを予測しました。しかし、このような空孔を多くもつ氷は、構造がもろく、従来の評価手法では安定性が正しく評価できないことも予測されていました。
本研究では、各結晶構造の熱力学的安定性だけでなく、力学的な安定性も評価し、より正確な負圧の氷の相図を作成しました。熱力学的には安定だと評価された多くの結晶構造が、温度とともに容易に崩壊し、結晶構造を維持できず、相図は低温では複雑、高温ほど単純になることがわかりました。また、負圧を極めて大きくしていくと、多くの低密度氷は崩壊し、氷lhや氷XVIのような比較的高密度で丈夫な構造が最後まで生き残るために、同じ相が相図上で2度以上出現するという、面白い現象が見付かりました。負圧という、実験ではほとんどアクセスできない条件でどんなことが起こりうるかを、シミュレーションにより予測した論文です。
The most stable phase in the negative pressure region (the stretched state of a substance) is the gas phase, but the crystal can also exist as a metastable state. In 2014, crystal ice XVI, which has a lower density than normal ice, was produced by a special process, and is expected to become stable under negative pressure. Therefore, attention is focused on finding new low-density ice. In our previous paper (MHYMT2017), we predicted that there are numerous crystal structures that are stable under negative pressure, and that the designed ice aeroice is stable over a very wide temperature and pressure range. However, it was also predicted that ice with many such pores would be fragile in structure, and the stability could not be correctly evaluated by conventional evaluation methods.
In this study, we evaluated not only the thermodynamic stability but also the mechanical stability of each crystal structure, and created a more accurate negative pressure ice phase diagram. It was found that many crystal structures evaluated to be thermodynamically stable collapse easily with temperature and cannot maintain the crystal structure, and that phase diagrams are complicated at low temperatures and simpler at high temperatures. At very high negative pressures, many low-density ice decays, and the same phase appears more than once on the phase diagram to allow relatively dense and robust structures such as ice lh and ice XVI to survive to the end. It is a paper that predicted what could happen under the condition of negative pressure, which is almost inaccessible in experiments, by simulation.